Introduction
Overcharge is one of the difficult to pass in the current lithium battery safety test, so it is necessary to understand the overcharge mechanism and the current measures to prevent overcharge.
Figure 1 shows the voltage and temperature curve of the NCM+LMO/Gr system battery when it is overcharged. The maximum voltage is reached at 5.4V, and then the voltage drops, eventually causing a thermal runaway. The voltage and temperature curves of the overcharged ternary battery are very similar to it.

When lithium batteries are overcharged, they will generate heat and gas, including ohmic heat and heat generated by side reactions, of which ohmic heat is the main. The battery side reaction caused by overcharge is first of all that excess lithium is embedded in the negative electrode, and lithium dendrites will grow on the surface of the negative electrode (the N/P ratio will affect the initial SOC of lithium dendrite growth). Secondly, excessive lithium is released from the positive electrode, causing the cathode structure to collapse, releasing heat and releasing oxygen. Oxygen will accelerate the decomposition of the electrolyte, and the internal pressure of the battery will continue to rise. To a certain extent, the safety valve will be opened. Exposure to active substances and air will further generate more heat. Studies have shown that reducing the number of electrolytes will significantly reduce heat and gas production during overcharge. Other studies have been studied that when the battery cannot be opened normally without a splint or the safety valve when overcharged, the battery is prone to explosion.
A slight overcharge will not cause thermal runaway, but it will cause capacity attenuation. The study found that when the NCM/LMO mixture is overcharged, the capacity of SOC below 120% does not significantly attenuate, and when the capacity of SOC is higher than 130%, it will attenuate significantly.
At present, there are several ways to solve the problem of overcharge:
1) Set a protective voltage in BMS, which is usually lower than the peak voltage when over‐charged;
2) Improve the anti-overcharge ability of the battery through material modification (such as material coating);
3) Add anti-duplicate additives to the electrolyte, such as redox pairs;
4) The use of voltage-sensitive membranes. When the battery is overcharged, the membrane resistance is significantly reduced and plays a shunt role.
5) OSD and CID design is used in square aluminum case batteries, which is currently a universal anti-charge design. Soft package batteries cannot achieve a similar design.
References
Buy Now: Semco Infratech – SI Battery Charge & Discharge Cabinet Tester – SI BCDS 100V 20A 4CH.
Energy Storage Materials 10 (2018) 246–267
This is a brief introduction to the changes in voltage and temperature of lithium cobaltate Batteries during overcharge. The figure below shows the overcharge voltage and temperature curve of the lithium cobaltate battery, and the horizontal axis is the amount of lithium removal. The negative electrode is graphite, and the electrolyte solvent is EC/DMC. The battery capacity is 1.5Ah. The charging current is 1.5A, and the temperature is the internal temperature of the battery.
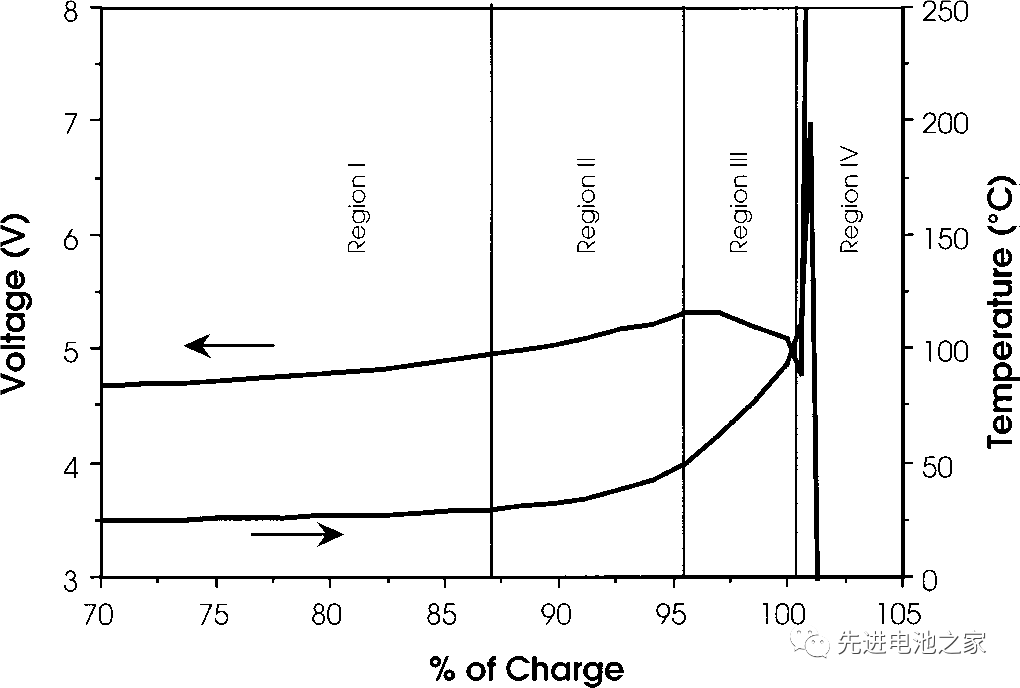
Lithium battery overcharge can be roughly divided into four areas, each of which is characterized as follows:
Area I
1. The battery voltage rises slowly. Lithium cobaltate is more than 60% DE lithium from the positive electrode, and lithium metal is precipitated on the negative side.
2. The battery is swollen, which may be due to the high-pressure oxidation of the electrolyte on the positive side.
3. The temperature is basically stable and rises slightly.
Area II
1. The temperature began to rise slowly.
2. In the range of 80-95%, the positive impedance increases, and the internal resistance of the battery increases, but decreases by 95%.
3. The battery voltage exceeds 5V, reaching the highest.
Area III
1. At about 95%, the battery temperature began to rise rapidly.
2. From about 95% until it approaches 100%, the battery voltage drops slightly.
3. When the internal temperature of the battery reaches about 100°C, the battery voltage drops sharply, which may be caused by the lower internal resistance of the battery due to the increase in temperature.
Area IV
1. The internal temperature of the battery is higher than 135°C, the PE diaphragm begins to melt, the internal resistance of the battery increases rapidly, the voltage reaches the upper limit (~12V), and the current drops to a lower value.
2. Between 10-12V, the battery voltage is unstable and the current fluctuates.
3. The internal temperature of the battery rises rapidly, and the temperature rises to 190-220°C before the battery breaks.
4. The battery is cracked. The overcharge of ternary batteries is similar to that of lithium cobaltate batteries. At present, when the ternary square aluminum case battery on the market is overcharged, it is roughly controlled to start OSD or CID when entering Zone III to cut off the current and protect the battery from overcharge.
References
Buy Now: Semco Infratech – SI Battery Management System Tester – SI BMST 1-32S 60/120A with cabinet.
Journal of the Electrochemical Society, 148 (8) A838-A844 (2001)
Lithium battery overcharge mechanism and anti-overcharge measures (3)
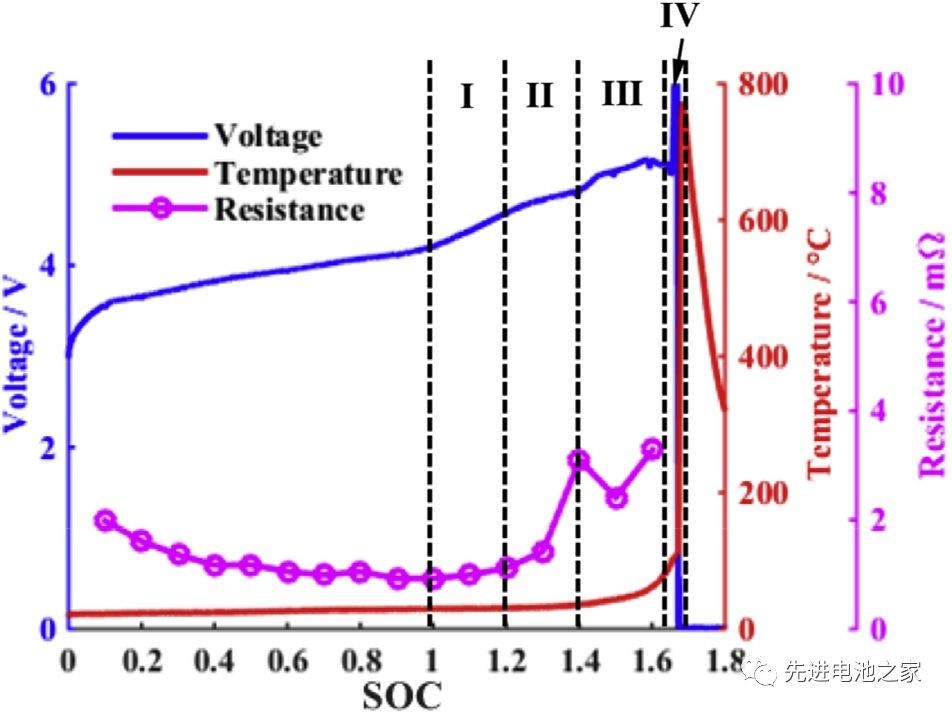
In this paper, the overcharging performance of a 40Ah soft package battery with a positiveNCM111+LMO is studied through experiments and simulation, with overcharging currents of 0.33C, 0.5C, and 1C respectively. The battery size is 240mm*150mm*14mm. According to the rated voltage of 3.65V, the volume ratio energy is about 290Wh/L, and the specific energy is still relatively low.)The changes in voltage, temperature and internal resistance during overcharge are shown in Figure 1. It can be roughly divided into four stages:
Stage 1: 1<SOC<1.2, there is no obvious side reaction inside the battery, and the battery temperature and internal resistance change less.
Stage 2: 1.2<SOC<1.4, Mn in the positive electrode is dissolved, oxidized in the positive side electrolyte, and lithium metal is precipitated on the negative electrode surface. The reaction of lithium metal with the solvent thickens the SEI film, increases the battery impedance, and the battery temperature begins to rise slowly.
Stage 3: 1.4<SOC<1.6, the battery temperature rises faster, the battery swells significantly, and the cathode electrolyte oxidation accelerates, releasing a large amount of heat and gas. Lithium metal on the negative electrode surface continues to precipitate, the SEI film begins to decompose, and lithium-based graphite reacts with the electrolyte. Due to the change in the structure of the cathode material, the battery voltage drops slightly after reaching a peak of 5.2V.
Stage 4: SOC>1.6, the internal pressure of the battery exceeds the limit, the shell breaks, The diaphragm shrinks and deforms, and the battery is thermally out of control. Short circuits occur inside the battery, a large amount of energy is quickly released, and the battery temperature rises sharply to 780°C.
The schematic diagram of the side effects of each stage of overcharge is shown in
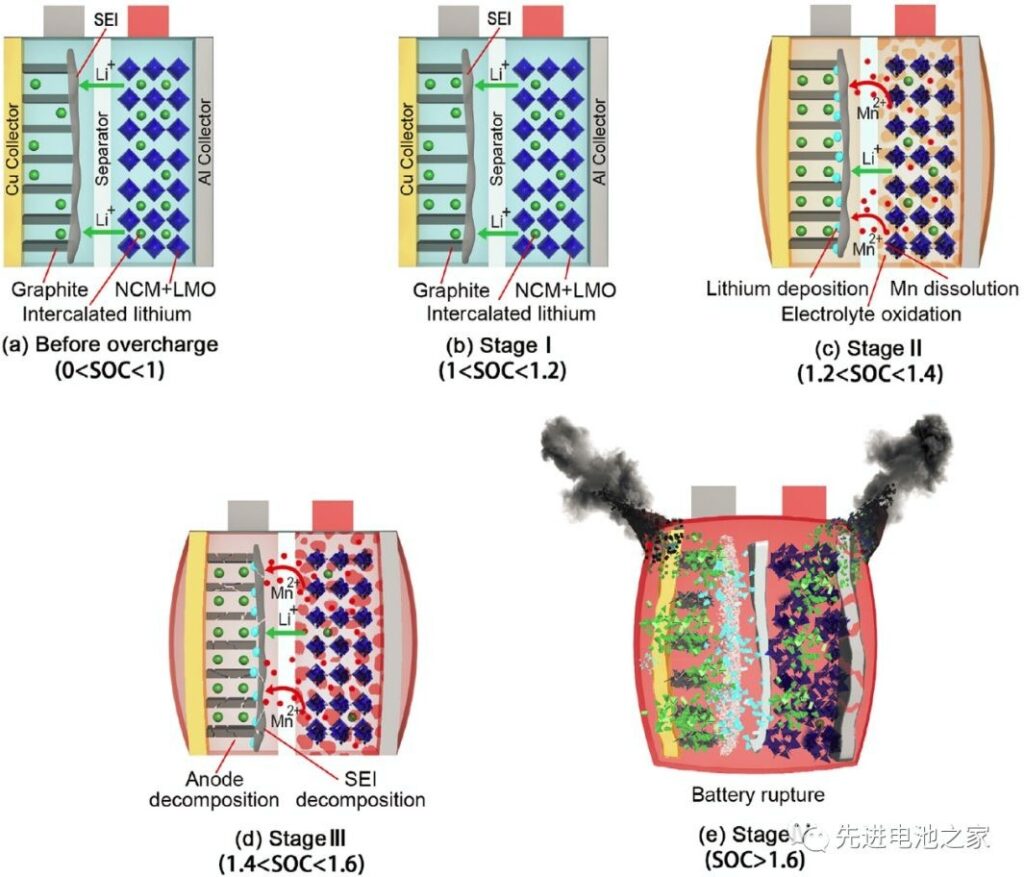
Figure 2
Heat production during overcharge includes reversible entropy heat, joule heat, chemical reaction heat, and heat released by an internal short circuit. Among them, the heat of chemical reaction includes Mn dissolution, lithium metal reaction with electrolyte, electrolyte oxidation, SEI membrane decomposition, negative decomposition, and heat released by positive electrode (NCM111 and LMO) decomposition. Table 1 shows the enthalpy and activation energy of each reaction. This paper ignores the side effects of binders).
Table 1
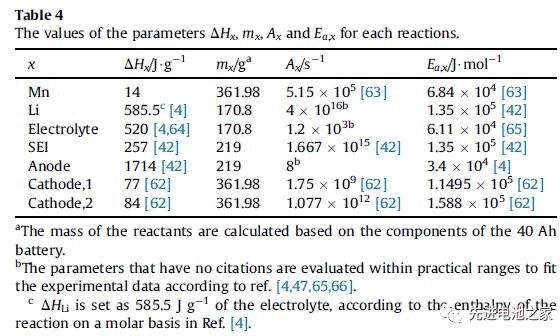
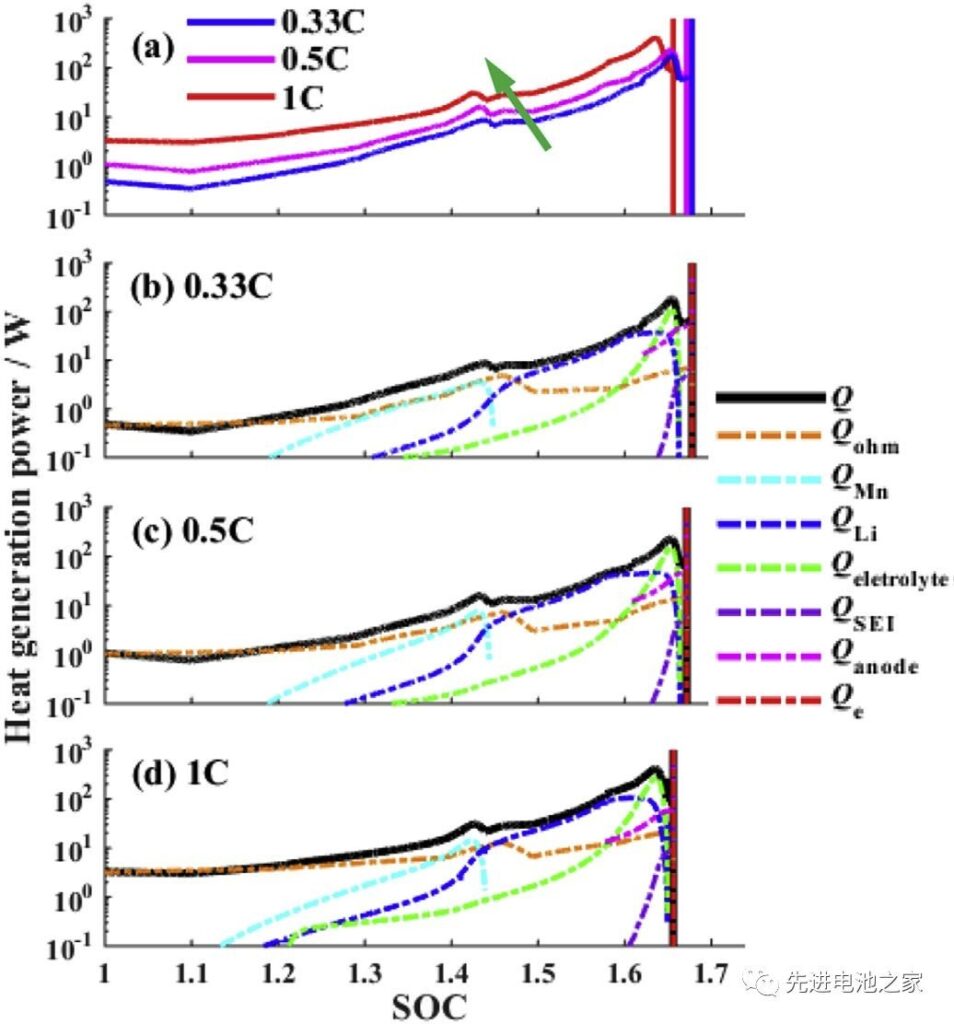
Figure 3 is a comparison of the heat production rate when different charging currents are overcharged. The following conclusions can be drawn from Figure 3:
1) With the increase of charging current, the thermal runaway time is advanced.
2) The heat produced in overcharge is mainly Joule fever. SOC<1.2, the total heat production is basically equal to Joule fever.
3) In the second stage (1<SOC<1.2), the reactions of Mn dissolution, lithium metal reaction with electrolyte, and electrolyte oxidation began one after another. At 1C current, the reaction will be advanced.
4) SOC>1.45, the heat released by the reaction of lithium metal with the electrolyte will exceed that of Joule.
5) SOC>1.6, the DECOMPOSITION OF THE SEI MEMBRANE AND THE NEGATIVE DE‐ COMPOSITION BEGINS, AND THE ESTIMATION RATE OF THE ELECTROLYTE Oxide‐ TION REACT INCREASES QUID QUID, AND THE TOTAL CYPERMAL PRODUCTION RATE REACHES PEAK. (The descriptions 4 and 5 in the literature do not match the picture. Here, the picture is subject to adjustment.)
6) During the overcharge process, the reaction between lithium metal and electrolyte and electrolyte oxidation are the main reactions.
Through the above analysis, the electrolyte oxidation potential, negative polar capacity, and thermal runaway starting temperature are the three key parameters of overcharge. Figure 4 shows the impact of three key parameters on overcharge performance. It can be seen that the improvement of the oxidation potential of the electrolyte can greatly improve the overcharge performance of the battery, while the negative capacity has little impact on the overcharge performance. In other words, high-voltage electrolyte helps to improve the overcharge performance of the battery, and increasing the N/P ratio has little impact on the overcharge performance of the battery.)
More Articles:
Factors Affecting The Performance of High Power Lithium-ion Batteries,
Electrical measurement of lithium-ion batteries,
Functions of the Battery management system (BMS),
Why do electric vehicles get slower and slower after riding,
why can’t Electric Cars of the same model overt beat,
what to do if the electric car is flooded,
Lithium Battery Pack Details,
BMS Function Analysis of Battery Management System,
Battery Vulcanization,
The Charge Status of Lithium Batteries.,
How to distinguish between good and bad electric vehicle tires,


