Introduction
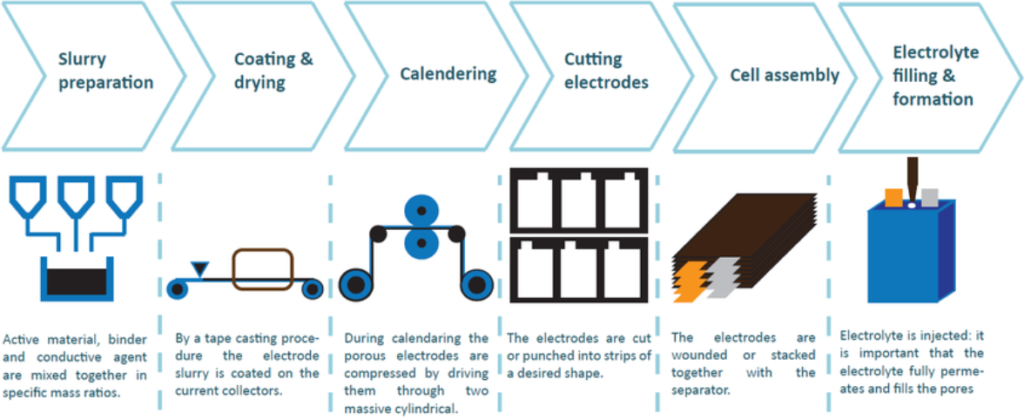
The production process of lithium-ion batteries is divided into four main processes: pole piece production, battery cell (cell) production, cell activation detection, and battery packaging. The production of pole pieces includes the processes of pulping, coating, rolling, slitting, sheet making, and tab forming. It is the basis of lithium-ion battery manufacturing and has high requirements for the performance, accuracy, stability, automation level, and production efficiency of pole-piece manufacturing equipment.
Safe operation of battery pack: –
Operator qualification-
- Know the professional knowledge of button electricity
- Have an electrician certificate and know the circuit knowledge
- Have undergone relevant professional training, and have the ability to identify possible hazards.
Power battery storage loading and unloading-
Loading and unloading-
- Use another car to lift the tray of the battery system off to the same plane as the hydraulic lifting platform and push the electric box to the hydraulic lifting table.
- Lift the vehicle with a lifting frame to the appropriate height- push the hydraulic lifting trolley under the car, adjust the direction of the electric box align the installation position, raise the hydraulic lifting platform and fine-tune the position of the lifting platform, align the positioning hole with the mounting hole and continue to raise the hydraulic lift after the table is aligned to each hole position, install it.
- Y connect the front and rear box connectors to complete the installation and connect with the high and low voltage of the vehicle. The wire connection is carried out inside the car body.

Storage and loading and unloading of power batteries-
Handling; –
Take it lightly, face up, not upside down or diagonally: –
- Use special means of transportation, it is strictly forbidden to move, drag or damage the appearance of the battery with bare hands. (For example, scratches and dents are caused).
- Check the insulation resistance before handling- total positive and total negative to the insulation resistance of the metal outer wall of the system 500MH.
- Check whether the relay is disconnected before handling- the front and rear box connectors should be protected.
- Storage temperature-0°c~25°c; humidity <75%R.H (anhydrous vapor condensation).
- The storage room is clean dry cool and ventilated to avoid contact with corrosive objects or Gas contacts.
- Clearly marked keep away from fire and heat sources and do a good job for insulation treatment. The protective cover of the battery box is installed and fastened without defects such as defects or damage.
- The positive and negative terminals are insulated with insulating tape or other insulating materials without gold.
- Short term storage- (within 3 months) SOC > 30% battery voltage >2.5 V.
- Long-term storage capacity >50% battery cell voltage >3.0V.
- Regular charge and discharge tests are required. It is recommended to charge and discharge once in a month (discharge current 0.2C and check the BMS function at the same time).
Buy Now: Semco Infratech – SI Battery Charge & Discharge Cabinet Tester – SI BCDS 100V 20A 4CH.
Outline
- Battery principle and development of chemical batteries
- Lithium-ion battery’s performance and safety
- Structure of ION phosphate button power battery
- Storage loading and unloading of power batteries
- Safe operation of the battery pack
Structure of ion phosphate button: –
Battery pack M-30
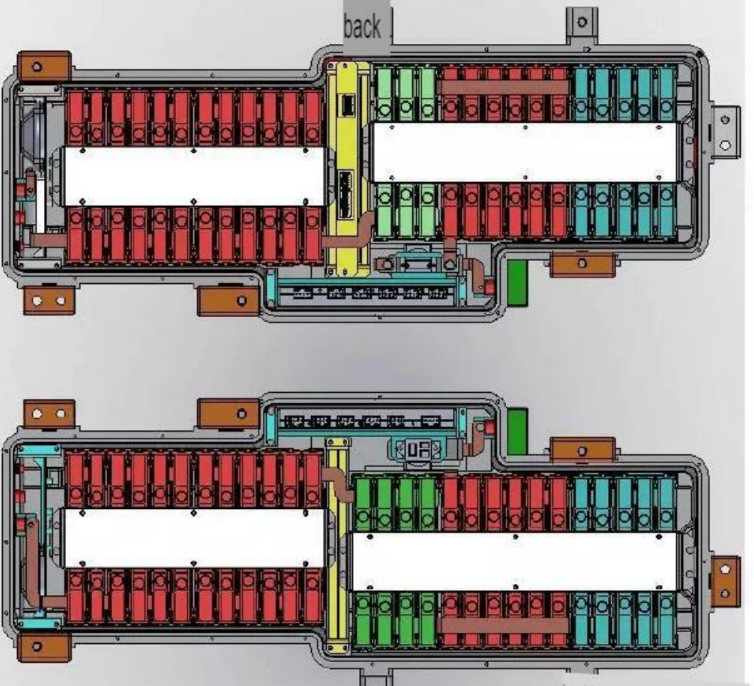
Ferric acid bond power battery structure: –
Cells and modules: –
A

Acquisition line
- Voltage acquisition
- Temperature collection

True iron –acid bond power structure-
Main components-
- Outer box-bracket function, box body, upper cover, an insulating film, pol column
- Y module –work unit battery cell, bus bar VBMS control system, BMU, voltage acquisition line, temperature acquisition
- Line relay switch
- Fuse safety protection
- Y-thermal management all-weather operation guarantee, heating plate, fan
- Other positive and negative leads, brackets
Key cell production
Coating, slitting, welding, top cover, pouring electrolyte.
The packet capacity is allowed to stand and become — let stand—encapsulation
Positive plate material of iron phosphate carp-
Iron phosphate button (LiFePo4, LFP for short)
A battery refers to a button-ion battery that uses iron phosphate buttons as the cathode material, and its working principle it is the same as lithium-ion batteries.
- The correct chemical formula of LiFePO4 (can be metal such as Fe, Co, MN, Ti, etc.)
- Its physical structure is an olivine structure. From the point of view of the structure, it can be used for ionization. The cathode material of the cell includes AyMPO4, Li 1, MFePO4, LiFePO4MO, etc. can be used as cathode materials.
- It is characterized by the fact that does not contain any heavy elements, the price of raw materials is low and the carbon iron and carbon dioxide are on the earth it is rich in resources, so there will be no major problems with feeding.
- In addition to the common characteristics of smart batteries, there are some unique advantages, such as their moderate operating voltage (3.2v) large capacity (170mAH/g) high discharge power can fast charging, and long cycle life (up to 2000 times) in high temperature and high heat environments with high stability.
Lithium-ion battery performance and safety: –
Over-discharge-
The voltage is placed at 2.0V the anode copper collector is dissolving in the electrolyte precipitates at the cathode and generates the copper dendrites to pierce the isolation film, causing a short circuit.
Heating-
Heating exceeds 150 degrees resulting in a thermal runaway (chemical energy, electrochemical energy, cathode structure field.
Low-temperature charging-
If the temperature is below 0 degrees, the battery is charged and it is expected to precipitate on the anode surface. The process is not possible.
The reverse reaction damages the battery.
E+E0-RT/nFlg [oxidation state /reduced state]
Lithium-ion battery performance and safety –
Smart ION battery design safety –
- Lithium-ion batteries are batteries that have been successfully commercialized and installed.
- Annual output of more than 2 billion.
- Overcharge and over-discharge, and low-temperature cycling caused damage.

Performance and safety of lithium-ion batteries-
Ion battery design safety-
- The inside of the V battery is almost vacuum and it can be charged and discharged in a confined environment.
- The Y diaphragm has a high-temperature protection function. When the temperature reaches 130° the ions cannot pass through and cut off the circuit.
- When the charging or discharging current is too large, the protection board inside the battery cell will cut off the current. Y is installed in the PTC temperature control terminal or fuse, which can limit the generation when the current is too large.
Key ION battery safety and performance-
Characteristics of ideal positive and negative electrode materials.
- It has a layered or multichannel crystal structure, which is conducive to the embedding and use of lithium-ion batteries.
- During the charging and discharging process, it is as easy and possible to embed and prolapse and it has high chemical properties and learning capacity.
- When fine ION are embedded and removed there is a relatively stable charge and discharge voltage.
- Wisdom IONs have a large diffusion coefficient, which reduced the energy consumption of ions when they move and protects proven good charge and discharge performance.
- The Embedded compounds should be cheap less polluting and light in quality.
Buy Now: Semco Infratech – SI Battery Management System Tester – SI BMST 1-32S 60/120A with cabinet.
Lithium-ion battery performance and safety –
Graphite anode material capacity
- At full charge: LIC 6<>6c+Li++e-
- 6C atomic energy emits 1 electron, and 6 moles of C atoms (72 grams) can emit
- 1 mole of electrons = 1.60217733*10, 19*6.022*1023
- =96485 Cullen
- =26.80 an hour
- Theoretical graphite gram capacity = 26.80/72=372mah
- Actual graphite gram capacity = 280~370mah
Key ion battery performance and safety
Comparison of material properties of a lithium-ION battery of cathode plate
| Material | Theoretical capacity (mah/gram) | actual gram capacity (mah/g) | Density (kg/l) | The volume energy density (ah/L) | voltage (V) | Security cost | Security cost |
| Lico 02 | 275 | 140 | 5.05 | 808 | 3.7 | ||
| lini 02 | 274 | 220 | 4.80 | 1056 | 3.4 | ||
| Limn 04 | 148 | 110 | 4.20 | 462 | 3.8 | good | low |
| LiFe P04 | 170 | 120-160 | 3.70 | 592 | 3.4 | Slow |
Based on safety and cost factors, the iron phosphate button is under the current technical level the best choice.
Lithium-ion battery performance and safety-
Electrolyte material composition-
Solvent – pc
- Ethyl stearate (EC)
- Dimethyl enanthate (DEC)
- Jiaxi
- 1,4 CBL
Solute – LiPF 6
- 1iHF 4
- LiCo 4
- LiAsFs
- LiTCF3SO3
- When using cool carbonate acid as a solvent, the flash point is low. it will flash ignite at a low temperature which is safe.
- Elephant generation solvents (including fish generation lipids) have a high flash point or even no flash point which is conducive to willow combustion of electrolyte. Oxygenators have less damage to battery performance and have an obvious effect on inhibiting electrolyte combustion. However, the use of oxide will greatly increase the production cost of a button–ion batteries.
- At present commercial lithium-ion, batteries use EC and DMC as solvents and LiPF2 as pinch salts which have high ionization properties sub conductive, and good electrochemical stability.
Lithium ION battery performance and safety-
Cathode material for lithium ION battery: LxMYoZ
- LiCo02
- VLini (1-x) CoxMn (1-x)/2O
- LiMnO4
- LiFePO4

Working principle-
When an external power and supply charges the battery the electron c on the positive electrode passes through the outside at this time Part of the circuit runs to the negative electrode from the positive electrode. “Climb over” the cooking hole in the diaphragm and swim to the negative electrode. Combine with the electronics that have already come over. When the battery has discharged the mechanism, it is just the opposite of charging. Taking LiFePO4 as an example it is a chemical; reaction.
The equation is-
Charging – LiFePO4 –xLi “-Xe”—xFePO4+ (1-x) LiFePO4
Discharge – FePO4 + xli *+ -Xe –xLiFePO4 + (1-x) LiFePO4
Performance and safety of ion-lithium batteries
Gaozi battery structure-
- Cathode-carp ion salt composed of transition metal oxide containing carp is commonly used. The material is diamond acid buttons fierce acid buttons ternary material and iron phosphate.
- Button-Anode: – Graphite graphitized carbon material, modify graphite, graphitized intermediate phase. Carbon particles, smart ION storage warehouse.
- Isolation film – a special composite film, whose function is to isolate positive and negative electrodes and prevent electrons pass through while allowing fine ions to pass through thus completing the rapid transfer of metal ions between the positive and negative electrodes during electrochemical charge and discharge loss. At present, it is mainly polyethylene (PE) or polypropylene (PP). Isolation anode and anode to prevent short circuit ion conduction and electronic.
- Isolation electrolyte – An organic electrolyte most of which is made of hexafluorophosphate carp (LiFL4) adds an organic solvent to make it a carp ion channel.
- Poles- Ni (CU), Al, electronic channel, external circuit a packaging film stainless steel shell, to maintain a stable electrochemical environment. Outer packing isolating film.
Li-ion battery ( Li-on battery ) Is currently the best energy storage solution at the same time it has both high power density and high energy density which is a current choice of an electric vehicle. The root cause of the sub-battery.
Lithium ion-battery and safety performance and safety-
The development of industrial society is increasingly dependent on portable electronic devices. The battery provided as the energy source of the equipment must be equipped with a higher energy density, longer cycle life, and safety. Plasma electricity commercialized in 1992 pool provides possibilities.
Main purpose-
- Electronic instrument
- Aviation power
- Consumer goods
- Electric vehicle moves

Outline-
- Battery principle and development of chemical batteries
- Smart ion- battery performance and safety
- The true ion-acid bond power battery structure
- Power battery storage loading and unloading
- Safe operation of the battery pack
Battery principle and development of chemical batteries-
Battery development-
The research and development of smart secondary batteries are divided into metal rechargeable secondary batteries. There are three stages of compound battery. In 1999, lithium-ion polymer batteries were officially put into operation for commercial production, and eight companies led by Panasonic launched products. So, 1999 was called the first year of carp polymer batteries by the Japanese. It uses carbon as the negative electrode. A compound containing carp is used as the positive electrode during the charging and discharging process there is no metal carp used as the positive electrode. During the charging and discharging process, there is no metal carp. There is only a pinch ion, which is the origin of the name of the pinch battery.
Battery principle and development of chemical batteries: –
Battery classification-
Rechargeable batteries (primary batteries), such as zinc dry batteries alkaline batteries, and silver batteries, secondary batteries (accumulators) such as lead-acid batteries, wire oxygen batteries, and button batteries. Fuel cells, hydrogen, methanol, methanol fuel cells.
Solar cell-
Basic parameters of common secondary batteries-
| Types of technical parameters | Battery | Battery | Liquid wisdom ion-battery | Polymer wisdom ion-battery |
| Operating voltage | 1.2-1.5 | 1.2-1.5 | 3.7 | 3.7 |
| Mass-specific energy (wh/kg) | 50 | 65 | 100-60 | 120-170 |
| Charge and discharge time | 500 | 500 | 500-1000 | >1000 |
| Self-discharge rate (%month) | 25-30 | 30-35 | 6-9 | 3 |
| Memory effect | Serious pollution Heavy metal pollution | None | Pollution free | Pollution free |
| Security | High | In | low | In |
Battery principle and development of chemical battery-
The basic composition of chemical battery: –
- Positive electrode
- Isolation film, wire outer packing
- Electrode reaction
- Discharge
Anode CO2 ++2e =Cu
Cathode ZN-2~=Zn2+
- Battery energy
Battery energy = potential difference * power consumption
- The necessary potential difference is determined by the potential of the positive and negative electrode materials.
- The total amount of electricity passing through the circuit.
Outline-
Battery principle and development of chemical batteries
- Lithium-ion battery performance and safety
- Structure of lithium iron phosphate battery power
- Power battery storage loading and uploading
- Safe operation of a battery pack
Conclusion
Lithium-Ion batteries are a great power source for projects but they require care during use and charging. They can be easy to damage or misuse and can hurt anyone. All batteries should be tested and certified but it should be used carefully.
More Articles:
The packaging form of lithium-ion battery is better,
Causes Reconditioning Of Battery Vulcanization,
Lithium-ion Battery Process Details,
Lithium battery overcharge mechanism and anti-overcharge measures.,
Factors Affecting The Performance of High Power Lithium-ion Batteries,
Electrical measurement of lithium-ion batteries,
Functions of the Battery management system (BMS),
Why do electric vehicles get slower and slower after riding,
why can’t Electric Cars of the same model overt beat,
what to do if the electric car is flooded,
Lithium Battery Pack Details,


