Introduction
If lots of lithium batteries that are bulging, it is possible that the voltage of your commonly used battery charger does not match the lithium-ion battery, and it is recommended to replace it. In addition, if the lithium-ion battery is not used for a long time, there will be a problem of bulging (Bulging Reasons for Lithium Batteries).
Lithium battery is a rechargeable battery, which mainly relies on the roundtrip embedding and deem bedding of lithium ions between the positive and negative electrodes to realize the storage and release of energy.

Analysis of the Basic Structure of Lithium Battery Pack: Main material composition of lithium battery: positive material, negative material, electrolyte, diaphragm (isolation material)
Buy Now: SEMCO Battery Charge & Discharge Cabinet Tester SI BCDS 100V 10/20A 7CH.
Simple Structure of Lithium Batteries
Positive Pole
In terms of battery weight composition, cathode materials account for a large proportion (usually 70% to 80%), because the performance of cathode materials directly affects the performance of lithium-ion batteries, and their cost also directly determines the battery cost. Positive materials ac‐ count for 30% to 40% of the cost of lithium-ion batteries, which also directly affects the energy density and performance of lithium battery packs.
Negative Pole
Negative material is composed of materials with lower potential than the positive electrode, and has high specific capacity and good reversibility of charge and discharge, thus maintaining good size and mechanical stability (no serious deformation) in the lithium insertion process. Negative electrode materials mainly affect the efficiency and circulation performance of lithium batteries, and the performance of negative materials also affects the performance of lithium batteries. Negative materials account for about 10-20% of the total cost of lithium batteries. The types of anode materials include carbon anode and non-carbon anode.

Electrolyte
Electrolyte acts as a transport charge between the positive and negative electrodes (similar to the carrier in radio) and has a high ion conductivity, which should generally reach 1×10-3~2×10-2 S/cm. It affects the energy density, cycle life, power density, safety performance and other factors of lithium battery packs.
Diaphragm
The diaphragm has a fixed pore diameter and porosity, which ensures low resistance and high ion conductivity, has good penetration for lithium ions, has good infiltration of electrolyte and sufficient suction and moisturizing ability to maintain ion conductivity. At the same time, it has electronic insulation to ensure mechanical isolation of positive and negative poles. In addition, there should be enough puncture. Mechanical properties such as strength and tensile strength, corrosion resistance of electrolyte and sufficient electrochemical stability. Power batteries require higher diaphragms, and usually use composite membranes
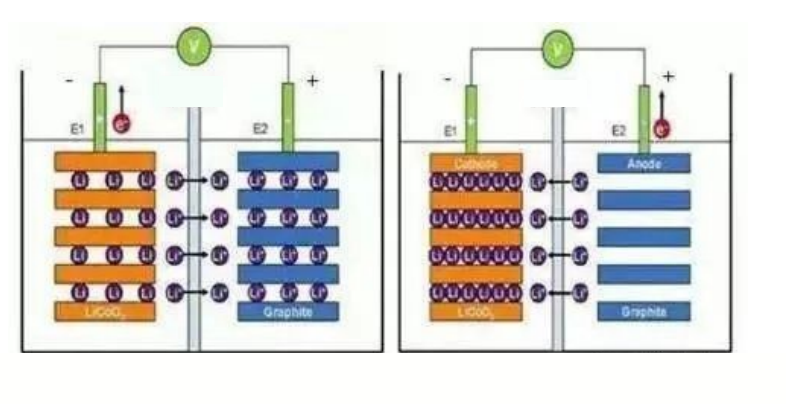
How lithium batteries work: Lithium batteries are rechargeable batteries, which mainly rely on the round-trip embedding and deem bedding of lithium ions between the positive and negative electrodes to realize the storage and release of energy
- Charging Process: Driven by the electric field, lithium ions are extracted from the positive lattice and embedded in the negative lattice through the electrolyte. If the voltage is less than 3V, it should be pre charged first. The charging current is 1/10 of the set current. After the voltage rises to 3V, it enters the standard charging process. The standard charging process is: constant cur‐ rent charging with a set current. When the battery voltage rises to 4.20V, it is changed to constant voltage charging, keeping the charging voltage at 4.20V. At this time, the charging current gradually decreases. When the current drops to 1/10 of the set charging current, the charge ends. This is a process of charging a lithium battery. If a lithium battery exists in a smart device, its charging mode will be controlled by smart device software
- Discharge Processes: the discharge process is exactly the opposite of the charging process. Lithium ions return to the positive electrode under the action of the electric field, and the electrons reach the positive electrode through the external circuit and are compounded with lithium ions. Battery discharge. At this time, the negative pole. The electron e on it runs from the external circuit to the positive electrode, and the positive lithium-ion Li+ jumps into the electrolyte from the negative electrode. Climb through the small winding hole in the diaphragm, “swimming” to the positive pole, combined with the electrons that have long run. By understanding the charging and discharging process of lithium batteries, we can understand microscopically that the capacity of batteries is actually the amount of charge contained in the battery.

Buy Now: SEMCO Battery Charge & Discharge Cabinet Tester SI BCDS 100V 10/20A 7CH.
Why does the battery bulge when it is used normally or not used for a long time?
There are Two Kinds of Bulging in the Process of Charging and Discharging:
1. Drums Caused by Over-Charge
Overcharging will cause all the lithium atoms in the cathode material to run into the negative material, causing the original full grid of the positive electrode to deform and collapse, which is also a major reason for the decline in lithium battery power: volume. In this process, there are more and more lithium ions at the negative electrode. Excessive accumulation causes lithium atoms to grow into tree stumps to crystallize, causing the battery to swell.
2. Drum Bag Caused by Over-Releasing
During the first charging and discharge of liquid lithium-ion batteries, the electrode material reacts with the electrolyte at the solid-liquid phase interface to form a passivation layer covering the surface of the electrode material. The formed passivation film can effectively prevent the passage of electrolyte molecules, but Li+ can be freely embedded and removed through the passivation layer, with the characteristics of solid electrolytes. Therefore, this passivation film is called solid electrolyte interface), abbreviated as SEI. The SEI film will protect the negative electrode material, so that the material structure is not easy to collapse, and can increase the cycle life of the electrode material. The SEI membrane is not invariant, and there will be slight changes in the process of charging and discharging, mainly because some organic matter will change reversibly. After the battery is over discharged, the SEI diaphragm will be reversibly damaged, and the negative material collapses after the SEI failure of the protective negative material, thus forming a bulging phenomenon
Conclusion
Bulging in lithium-ion batteries could prove to be dangerous and a potential risk to the users. Bulging of lithium-ion batteries occur due to too much current inside a cell of the battery causing a build-up of heat and gas. This can happen due to a lot of factors. Some of them are overcharging, damage to the battery, deep discharge state, or a manufacturer defect.
With the number of potential dangers of a swollen lithium-ion battery, it makes sense to get acquainted to a few basic tips to ensure that you and your devices are safe and out of danger.
- Don’t leave your lithium-ion battery powered device plugged in unnecessarily.
- Always use proper power charger.
- It makes sense to quickly replace the lithium-ion battery if it is damaged or is no longer offering the specified performance.
- It makes sense to always store your smartphone device in a cool, dry environment. Remember, high-temperature may cause damages to the lithium-ion battery.
More Articles:
Aging of NCA Lithium Batteries,
Function and Components of Battery Pack & BMS,
Cell BMS Pack Failure Analysis,
Key Points of Lithium-Ion Battery Production Process Control,
Lithium-ion Battery Packaging Information for Beginners,
Lithium Battery Protection Board,
Basic Knowledge of Lithium-ion Battery Commercialization,
Action protector in electric vehicle charging system,
Production problems of lithium batteries,
How the Three Electrode System Works,
Electric Vehicle Maintenance Practical Dry Goods,


